Gas Pressure and Manometer
Quiz Summary
0 of 12 Questions completed
Questions:
Information
You have already completed the quiz before. Hence you can not start it again.
Quiz is loading…
You must sign in or sign up to start the quiz.
You must first complete the following:
Results
Results
0 of 12 Questions answered correctly
Your time:
Time has elapsed
You have reached 0 of 0 point(s), (0)
Earned Point(s): 0 of 0, (0)
0 Essay(s) Pending (Possible Point(s): 0)
Categories
- Not categorized 0%
| Pos. | Name | Entered on | Points | Result |
|---|---|---|---|---|
| Table is loading | ||||
| No data available | ||||
- 1
- 2
- 3
- 4
- 5
- 6
- 7
- 8
- 9
- 10
- 11
- 12
- Current
- Review
- Answered
- Correct
- Incorrect
-
Question 1 of 12
1. Question
1 point(s)A water manometer is used to measure the pressure of a gas supply to a house. It gives a reading of \(h \mathrm{~cm}\) of water.
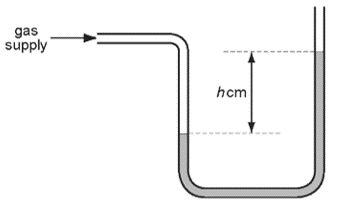 Why is it better to use water rather than mercury in this manometer?CorrectIncorrect
Why is it better to use water rather than mercury in this manometer?CorrectIncorrect -
Question 2 of 12
2. Question
1 point(s)The relationship between pressure \(p\) and volume \(V\) of a gas is given as \(p V=\) constant.
Under which conditions for the mass of a gas and for its temperature does the equation hold?\[
\begin{array}{|c|c|c|}
\hline & \text { mass } & \text { temperature } \\
\hline \text { A } & \text { changing } & \text { changing } \\
\text { B } & \text { changing } & \text { constant } \\
\text { C } & \text { constant } & \text { changing } \\
\text { D } & \text { constant } & \text { constant } \\
\hline
\end{array}
\]CorrectIncorrect -
Question 3 of 12
3. Question
1 point(s)A manometer is used to measure the pressure of a gas supply.
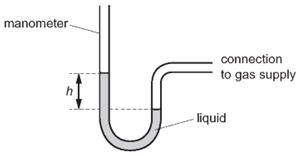
Which change gives a greater value of height \(h\) ?CorrectIncorrect -
Question 4 of 12
4. Question
1 point(s)The volume of a gas is measured at different pressures.
The pressure \(p\) and the volume \(V\) of the gas are found to be related by the equation:
\[
p V=\text { constant }
\]
Which quantities are kept constant and which quantities change?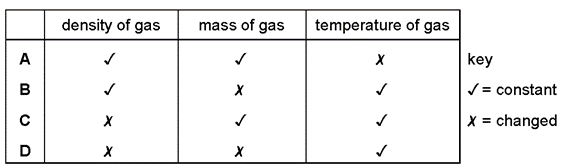 CorrectIncorrect
CorrectIncorrect -
Question 5 of 12
5. Question
1 point(s)A diver under water uses breathing apparatus at a depth where the pressure is \(1.25 \times 10^5 \mathrm{~Pa}\).
A bubble of gas breathed out by the diver has a volume of \(20 \mathrm{~cm}^3\) when it is released. The bubble moves upwards to the surface of the water.
At the surface of the water, the atmospheric pressure is \(1.00 \times 10^5 \mathrm{~Pa}\).
The temperature of the water is the same at all depths.
What is the volume of this bubble when it reaches the surface?CorrectIncorrect -
Question 6 of 12
6. Question
1 point(s)The diagram shows a manometer with one side connected to a gas cylinder and the other side open to the atmosphere.
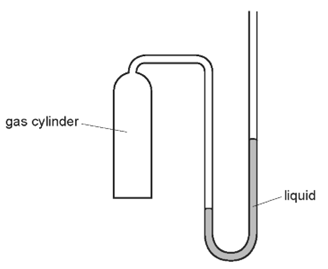
Which conclusion can be made using only the information from liquid levels in the manometer?CorrectIncorrect -
Question 7 of 12
7. Question
1 point(s)The volume of a gas in a sealed syringe is increased. The temperature of the gas does not change.
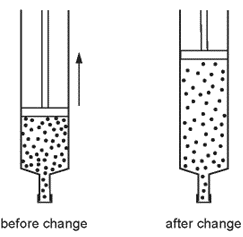
After this change is made, what has happened to the gas molecules in the syringe?CorrectIncorrect -
Question 8 of 12
8. Question
1 point(s)A manometer is used to measure the pressure of the air in a container.
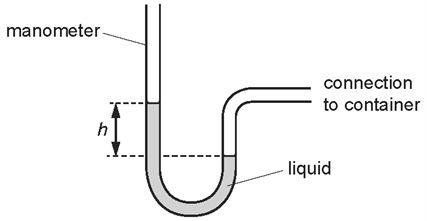
Which change would give a bigger value of height \(h ?\)CorrectIncorrect -
Question 9 of 12
9. Question
1 point(s)A student places his thumb firmly on the outlet of a bicycle pump, to stop the air coming out.
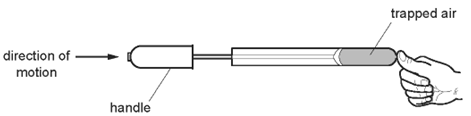
What happens to the pressure and what happens to the volume of the trapped air as the pump handle is pushed in?\[
\begin{array}{|c|c|c|}
\hline & \text { pressure } & \text { volume } \\
\hline \text { A } & \text { decreases } & \text { decreases } \\
\text { B } & \text { decreases } & \text { remains the same } \\
\text { C } & \text { increases } & \text { decreases } \\
\text { D } & \text { increases } & \text { remains the same } \\
\hline
\end{array}
\]CorrectIncorrect -
Question 10 of 12
10. Question
1 point(s)The diagram shows a mercury manometer used to measure the pressure of gas in a container. Atmospheric pressure is \(76 \mathrm{~cm}\) of mercury.
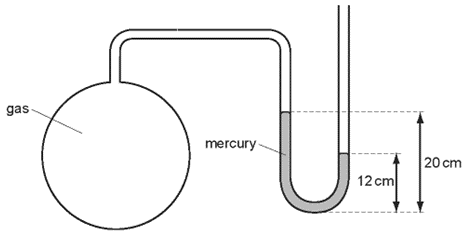
What is the pressure of the gas?CorrectIncorrect -
Question 11 of 12
11. Question
1 point(s)The pressure of a fixed mass of gas in a cylinder is measured. The volume of the gas in the cylinder is then slowly decreased. The temperature of the gas does not change.
Which graph could show the change of pressure of the gas during this process?CorrectIncorrect -
Question 12 of 12
12. Question
1 point(s)A water manometer is used to measure the pressure of a gas supply.
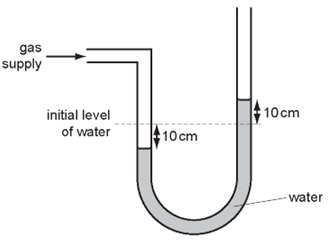
When it is attached to the gas supply, the water falls on the left side and rises on the right side. The difference in the levels of water on the two sides is now \(20 \mathrm{~cm}\).
What is the pressure of the gas supply?CorrectIncorrect